THE MISSING MECHANISM: How One Biological Failure Explains the Modern Neurodivergent Spectrum
If the system can respond, the system is not broken. And if it’s not broken... it can be rebuilt.

**THE MISSING MECHANISM:
How One Biological Failure Explains the Modern Neurodivergent Spectrum**
For more than a century, medicine has carved autism, ADHD, schizophrenia, bipolar disorder, Parkinson’s, and Alzheimer’s into separate kingdoms — different doctors, different clinics, different explanations.
But the data never truly fit that story. The same inflammatory markers. The same sleep signatures. The same metabolic scars. The same regional debris piling up in different decades of life. And then the papers of 2024–2025 landed, and the pattern snapped into focus with the force of a trap closing: these conditions are not separate illnesses. They are time-stamped expressions of one upstream failure — the collapse of the brain’s night-shift, the deep-sleep clearance system that keeps neural tissue safe. We didn’t discover a new disease. We discovered the mechanism tying them together.
The Night Shift Hypothesis
A unified mechanism emerging across autism, ADHD, schizophrenia, bipolar disorder, Parkinson’s, and Alzheimer’s
In the quiet hours of scientific history, paradigm shifts often arrive not with thunder but with the soft click of scattered pieces aligning into a single, inevitable shape. For over a century and a half—since Louis Pasteur and Antoine Béchamp sparred over germs versus terrain—we have debated the roots of disease in silos.
Germ theory triumphed, giving us antibiotics and vaccines, but it left the body’s internal landscape—the “terrain”—as a footnote, a vague notion dismissed by specialists tunneling deeper into their own domains.
Neurologists chased proteins in the brain. Psychiatrists balanced neurotransmitters. Gastroenterologists mended leaky guts. Sleep researchers mapped waves on EEGs. Immunologists hunted rogue cells.
Each group amassed data, published papers, and built careers on fragments that explained symptoms but never the synchronization: why these disorders exploded together in the same era, across the same societies, following the same upward curve.
The answer, it turns out, may lie in a mechanism so fundamental, so hydraulically mundane, that its absence escaped notice amid the clamor of molecular hunts.
It is not a gene, not a toxin, not a single pathogen.
It is the failure of the brain’s nightly waste removal—a “night shift” that clears the detritus of daily neural activity.
When this shift is canceled, the brain marinates in its own refuse, triggering a cascade of inflammation that reshapes cognition, behavior, and structure.
The symptoms vary by age and brain region, earning different diagnostic labels, but the upstream breakdown is identical.
This hypothesis—let’s call it the Night Shift Hypothesis—does not claim to explain every case or every nuance.
It is a model, testable and falsifiable, drawing from thousands of studies that, when overlaid, reveal a coherence we missed because no one was looking for a continent while mapping islands.
It honors the terrain:
the body’s ecosystem, disrupted by modern assaults, leading to a chain reaction that ends in the brain’s silent drowning. And it offers a path forward—not a cure-all, but a reversal of the sequence, grounded in biology rather than blame.
The Twin Signatures: Sleep and Clearance
At the heart of the model are two interlocking failures, observed without exception across the spectrum of these disorders.
First, the erosion of slow-wave sleep (SWS), also known as deep sleep or stage N3. This is not the light doze of early night or the vivid dreaming of REM; it is the profound, almost comatose descent where brainwaves slow to delta rhythms (0.5–4 Hz), body temperature drops, and repair processes dominate.
In a healthy night, SWS occupies 20–25% of total sleep, concentrated in the first half, lasting 20–40 minutes per cycle. It is the phase where growth hormone surges, muscles rebuild, and memories consolidate—but crucially, it is the only window for the brain’s deep cleaning.
Studies converge here with eerie consistency. Autistic children show 30–50% less SWS than neurotypical peers, often fragmented by arousals.
- ADHD correlates with delayed sleep onset and reduced delta power.
- Schizophrenia patients exhibit SWS deficits of up to 40%, predating psychosis by years.
- Bipolar disorder features unstable SWS architecture, worsening during manic phases.
- Parkinson’s erodes SWS early, accelerating alpha-synuclein buildup.
- Alzheimer’s brains lose SWS progressively, with amyloid plaques correlating inversely with delta wave duration.
This is not correlation; it is pathogenesis.
Longitudinal data from cohorts like the Framingham Heart Study and the Wisconsin Sleep Cohort show that SWS loss predicts cognitive decline a decade later.
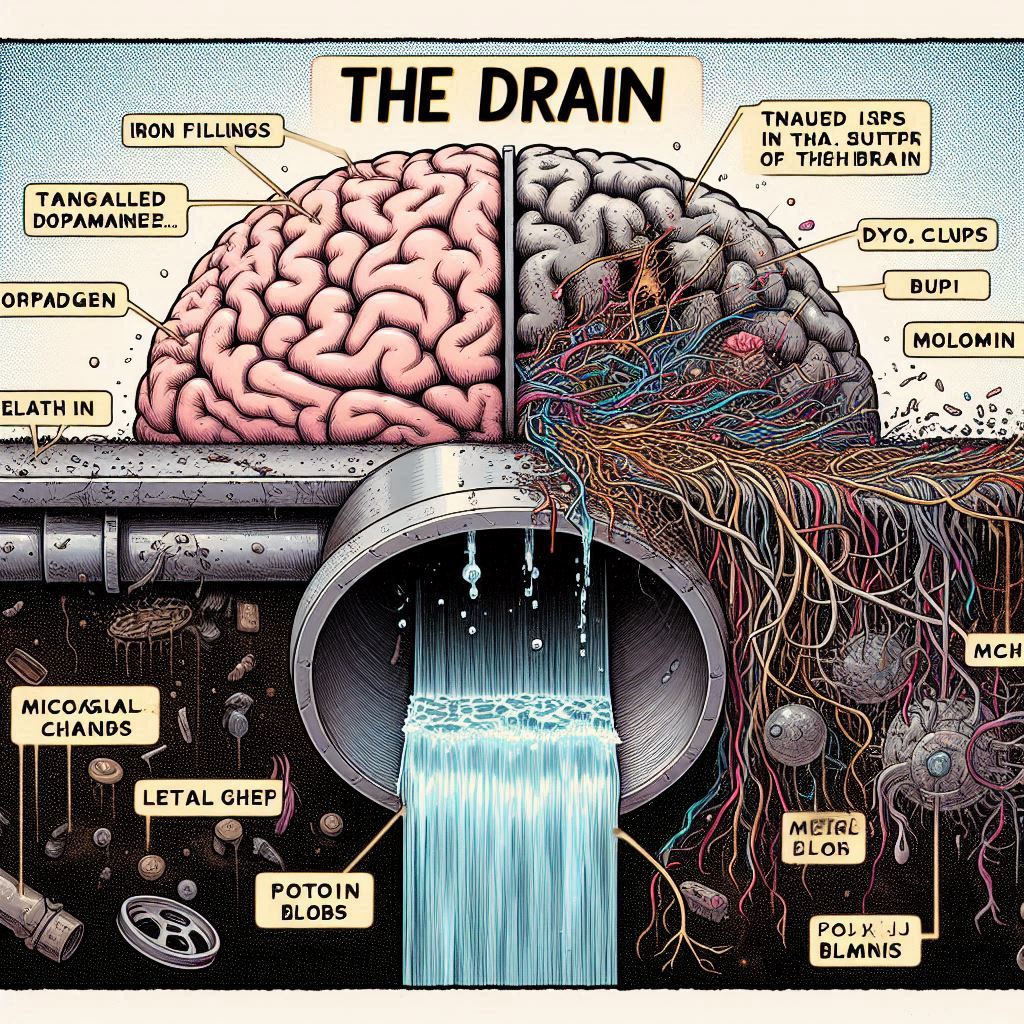
The second signature is the collapse of the glymphatic system, the brain’s waste-clearance pathway.
Discovered only in 2012 by Maiken Nedergaard’s team at the University of Rochester, it upended our view of the brain as an isolated fortress.
Unlike the body’s lymphatic network, which drains toxins via vessels, the brain relies on a perivascular “glymphatic” flow (a portmanteau of glia and lymphatics).
During SWS, noradrenergic tone drops, astrocytes (star-shaped support cells) shrink by 60%, expanding interstitial space. Aquaporin-4 channels on astrocytic endfeet align like floodgates.
Arterial pulsations drive cerebrospinal fluid (CSF) into the brain parenchyma, mixing with interstitial fluid and flushing waste—
>amyloid-β, tau, alpha-synuclein, oxidized dopamine metabolites, lipopolysaccharide (LPS) fragments from gut bacteria, labile iron, trace metals like aluminum and mercury, excitotoxins like quinolinic acid, and cellular debris—out through venous perivascular spaces and into meningeal lymphatics, eventually dumping into deep cervical lymph nodes.
Imagine a bustling city at night:
During the day, streets fill with trash from constant activity. Come midnight, traffic halts, sidewalks widen (astrocytes shrink), and pressure-washers (arterial pulses) flood the alleys with water (CSF), sweeping garbage into storm drains (meningeal lymphatics).
Without this nightly ritual, the city wakes to accumulating filth—rats multiply, structures corrode, residents sicken. In the brain, that filth is neurotoxic.
PET scans and CSF biomarkers confirm: glymphatic impairment precedes symptoms in every disorder mentioned.
- In autism, reduced clearance correlates with hippocampal metal accumulation.
- In schizophrenia, it explains oxidized dopamine buildup in the striatum.
- In Alzheimer’s, it accounts for 70–80% of amyloid retention.
These signatures are not side effects; they are the engine. But what cancels the night shift?
The Upstream Collapse: Terrain in the Gut
The chain begins not in the brain but in the colon, in a translucent gel layer we rarely discuss: the mucus bilayer.
This 200-micrometer barrier, renewed every few hours, is the terrain’s frontline—a semi-permeable shield separating trillions of gut microbes from the immune system.
In pre-industrial diets, it thrived on fibre, polyphenols, and microbial harmony.
Today, it erodes under assault: antibiotics wipe out communities; emulsifiers in processed foods dissolve it like detergent; glyphosate (Roundup) inhibits bacterial enzymes; seed oils promote oxidative stress; low-fibre diets starve its builders.
Central to this erosion are three keystone bacteria THE TRIO —Akkermansia muciniphila, Faecalibacterium prausnitzii, and Roseburia species—that form an interdependent triad.
Akkermansia degrades mucin for energy, regenerating the layer while producing short-chain fatty acids (SCFAs) like butyrate. Faecalibacterium consumes those SCFAs to suppress inflammation via IL-10 and Treg cells. Roseburia ferments fibres into butyrate, reinforcing the barrier and modulating immunity.
They are an ecosystem: lose one, and the others falter.
Metagenomic studies show their depletion in 80–90% of cases across
- autism (e.g., the ABIDE cohort), ADHD (Pediatric Longitudinal Cohort),
- schizophrenia (Gut-Brain Axis Project),
- neurodegenerative diseases (Parkinson’s Progression Markers Initiative).
Analogy: Think of the mucus layer as a coral reef. Akkermansia is the coral builder, Faecalibacterium the algae that feeds it, Roseburia the fish that cleans and protects. Strip the reef with pollution (modern diet), and the ecosystem crashes—waves crash through, eroding the shore (intestinal barrier), flooding the mainland (systemic inflammation).
With the Trio gone, two critical failures ensue.
First, gut permeability increases(leaky gut), allowing LPS (endotoxin from bacterial cell walls) to leak into circulation. LPS binds TLR4 receptors, activating NF-κB and cytokine storms—low-grade inflammation that signals “danger” to the brain via the vagus or bloodstream.
Second, choline metabolism collapses. The gut microbiome recycles 40–50% of choline (from diet and bile) via cutC enzymes in these bacteria. Choline is the precursor to acetylcholine, the parasympathetic neurotransmitter. Levels drop; synthesis falters.
The Master Switch: Vagus and the Descent into Chaos
Acetylcholine is the vagus nerve’s currency. This cranial nerve X, the longest in the body, is the autonomic highway from gut to brainstem, modulating heart rate, digestion, and—crucially—the shift from sympathetic (fight-flight) to parasympathetic (rest-digest-repair) dominance. Vagal tone, measured by heart rate variability (HRV), is acetylcholine-dependent.
When it falls, the body stays wired: cortisol lingers, noradrenaline surges, sleep fragments.
Without robust vagal signaling, SWS cannot initiate properly.
The brainstem’s nucleus ambiguus fails to quiet the locus coeruleus (noradrenaline hub), preventing the delta-wave descent.
Analogy: The vagus is the dimmer switch in a theater. Acetylcholine turns down the lights for the main show (SWS). Without it, the house lights flicker, the audience (neurons) never settles, and the performance (clearance) is canceled.
This loop amplifies: fragmented sleep worsens gut dysbiosis (via stress hormones), further depleting the triad, dropping acetylcholine more. Inflammation from LPS keeps microglia primed. The brain’s terrain becomes hostile.
Downstream: Waste, Inflammation, and the Spectrum of Suffering
Debris accumulates regionally. In young brains, hippocampal and amygdala overload manifests as sensory hypersensitivity and social withdrawal (autism).
- Prefrontal buildup disrupts executive function (ADHD).
- Limbic dopamine oxidation fuels hallucinations (schizophrenia).
- Nigral iron aggregation kills motor neurons (Parkinson’s).
- Cortical protein tangles erode memory (Alzheimer’s).
Microglia, sensing perpetual danger, polarize to M1 (pro-inflammatory) via pathways like NLRP3 inflammasome activation. They release TNF-α, IL-1β, and ROS, pruning synapses excessively (as in autism’s over-pruned networks) and perpetuating the cycle.
The Overlooked Clues
- Nicotine’s role: As a nAChR agonist, it mimics acetylcholine, boosting vagal tone and glymphatic flow temporarily. Smoking rates soar because it quiets the storm—self-medication for a cholinergic deficit.
- Heavy metals: Not primary causes, but trapped evidence. Glymphatic failure retains them in vulnerable regions, as MRI studies confirm.
- Suramin: This century-old drug blocks P2X/P2Y receptors, silencing ATP “danger” signals. Microglia relax to M2 (repair) mode, yielding weeks of symptom relief—until waste reaccumulates.
Reversing the River: Restoration and Implications
The sequence reverses upstream: Restore the Trio with inulin, resistant starch, fermented foods, polyphenols (e.g., beet kvass for betaine).
Mucus thickens; choline recirculates; acetylcholine rises; vagus reactivates via HRV practices (breathing, cold exposure).
SWS lengthens; glymphatics engage; microglia repolarize.
Pilot studies (e.g., FMT in autism) show promise: 50% SWS increase correlates with behavioral gains. This is terrain medicine—holistic, preventive, challenging silos.
The shift? It ends blame games, uniting fields.
No more “autism genes” versus “vaccine triggers”—it’s ecosystem collapse, fixable. If validated, it reframes psychiatry as neurology, neurology as immunology, immunology as microbiology.
We did not lack evidence. We lacked synthesis.
The night shift was always there, waiting for us to notice its absence.
(Technical appendix with references on the science page.)
